Plant Signalling
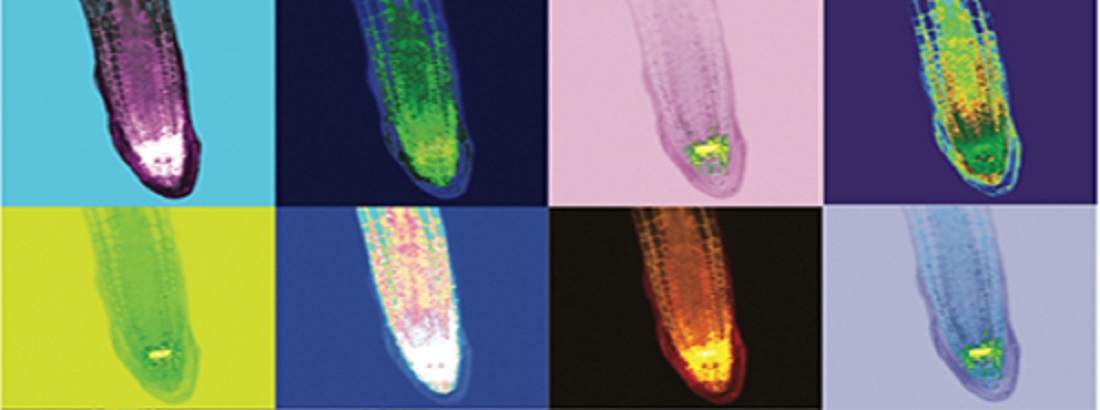
Signal perception at the cell surface by receptors and transduction of these signals to the cell interior is essential for plants to react and adapt to changes in the surroundings.
Studies of signal transduction and receptor activation that together modulate growth and development is a complex, but fascinating topic. This work requires a combination of biochemical, molecular and bioimaging methods.
Finally, we identify compounds that modulates both plant and fungal plasma membrane (PM) H+-ATPases as these have potential as growth regulators and drugs. We have recently performed a systematic screening of fungal compounds and determined their influence on the enzymatic activity of plant PM H+-ATPases. This have led to identification of several compounds that have applied potential as plant biologicals and can be produced at a large scale using established biotechnological techniques.
Find more information about the group in the menu below.
- Studies on how and what modulates plasma membrane H+-ATPase activity, a cellular element essential to growth.
- Providing systems for screening and identification of new signalling peptides, exploring new growth regulators and drug candidates.
- Design of synthetic organisms for peptide production, making use of biotech constructions for heterologous expression.
- Peptide mimics of plant pests, are they used for “tricking plant cells”?
For students wishing to work within these topics, M.Sc. and B.Sc. projects are currently available.
Please contact Anja Fuglsang atf@plen.ku.dk
De Novo Minibinders: De novo Minibinders and Cyclic Peptides as Anti-Infectives and Anti-Virulence Strategies Against Pathogenic Fungi. We provide a new discovery and evaluation platform for novel antimicrobial drugs.
Developing plant peptide hormones as biologicals to increase crop yield. A Novo Nordisk Foundation Pioneer Innovator project.
EcoPep: Pioneering Plant peptide hormones for sustainable agriculture. An Innovation Fund Denmark InnoExplorer project.
Live detection of secreted plant peptides. An Independent Research Fund Denmark, fields of ‘Technology and Production Sciences (FTP)’ project.
Fungaffine drugs: Exploring high-affinity peptides as anti-fungal agents. An Independent Research Fund Denmark, fields of ‘Technology and Production Sciences (FTP)’ project.
PlantsGoImmune.A new paradigm for disease-free crops of tomorrow. A Novo Nordisk Foundation Challenge project.
Precise, autocatalytic chemistry on proteins and peptides. Partner receiving grant provided by the Independent Research Fund Denmark, as a ‘Technology and Production Sciences (FTP)’ project.
Screening for modulators of H+-ATPase activity:
The mechanism behind tenuazonic acid-mediated inhibition of plant plasma membrane H+-ATPase and plant growth. Havshøi NW, Nielsen J, Fuglsang AT. Journal of Biological Chemistry 2024.
Trichoderma harzianum peptaibols stimulate plant plasma membrane H+-ATPase activity. Bjørk PK, Johansen NT, Havshøi NW, Rasmussen SA, Ipsen JØ, Isbrandt T, Larsen TO, Fuglsang AT. ACS Omega 2023.
Peptide-Receptor signalling:
Which came first – the messenger, the activator, or the receiver? Tost AS, Tidemand FG, Fuglsang AT. Trends in Plant Science 2026.
Evidence for multiple receptors mediating RALF-triggered Ca2+ signaling and proton pump inhibition. Gjetting SK, Mahmood K, Shabala L, Kristensen A, Shabala S, Palmgren M, Fuglsang AT. Plant Journal 2020.
Activation of the LRR Receptor-Like Kinase PSY1R Requires Transphosphorylation of Residues in the Activation Loop. Oehlenschlæger CB, Gersby LBA, Ahsan N, Pedersen JT, Kristensen A, Solakova TV, Thelen JJ, Fuglsang AT. Frontiers in Plant Science 2017.
Reviews:
A critical review on natural compounds interacting with the plant plasma membrane H+-ATPase and their potential as biologicals in agriculture. Havshøi NW, Fuglsang AT. Journal of Integrative Plant Biology 2022.
Proton and calcium pumping P-type ATPases and their regulation of plant responses to the environment. Fuglsang AT, Palmgren M. Plant Physiology 2020.
Collaborations:
Poltergeist-Like 2 (PLL2)-dependent activation of herbivore defence distinguishes systemin from other immune signalling pathways. Li R, Wang X, Ahmad FH, Fuglsang AT, Steppuhn A, Stintzi A, Schaller A. Nature Plants 2025.
Group members
| Name | Title | Phone | |
|---|---|---|---|
| Amalie Scheel Tost | Research Assistant | ||
| Anja Thoe Fuglsang | Professor | +4535332586 | |
| Frederik Grønbæk Tidemand | Assistant Professor | ||
| Gustav Blichfeldt Pedersen | Postdoc | +4535321297 | |
| Gustav Søndergaard Nymand | PhD Fellow | +4535331019 | |
| Hannah Bjerring Kastrup | Research Assistant | +4535329307 | |
| Marzanna Due | Laboratory Technician | +4535328186 |
Master's students
- Ida Engstrøm Garbers
- Katrine Johanne A Christensen
- Malte Bech Holde
- Maria Kristoffersen
- Mikkel Simoni Jørgensen
- Victor Severin Andersen
- Zhaopeng Lyu
Bachelor students
- Julius Leonardo Thougaard
- Mohamed Mehyar Abd Allah
- Regitze Fagerheim Lajer
Biosolutions student projects
- Marina Lopez Alonso


